Company Profile
QIAGEN is the leading global provider of Sample to Insight solutions that enable customers to gain valuable molecular insights from samples containing the building blocks of life. QIAGEN provides solutions to more than 500,000 customers around the world in Molecular Diagnostics (human healthcare) and Life Sciences (academia, pharma R&D and industrial applications, primarily forensics). QIAGEN currently has more than 5600 employees based in more than 35 countries and with sales topping $1.87 BN in 2020.
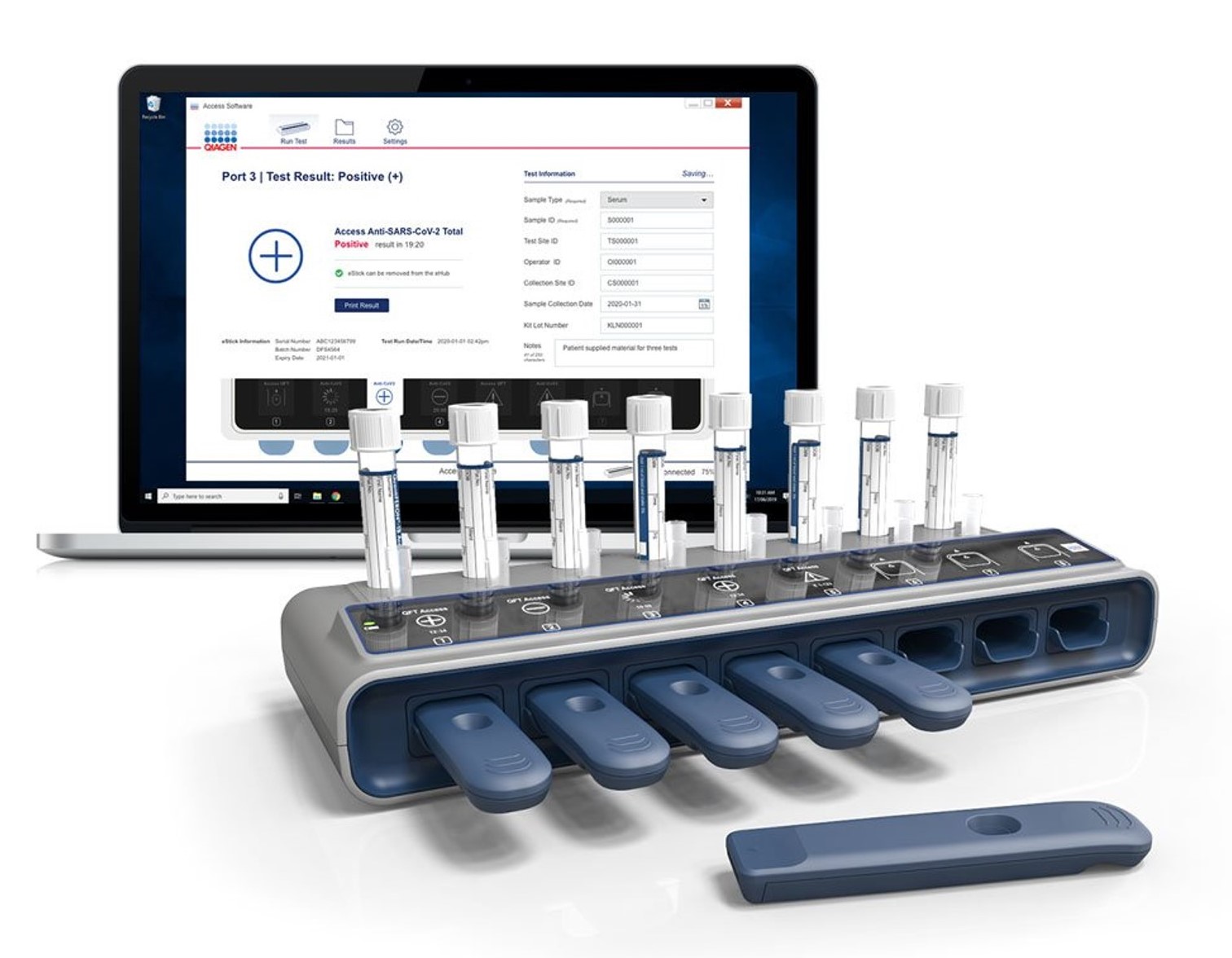
In 2020, QIAGEN announced its plans to launch an easy to use and cost effective rapid test for the detection of SARS-CoV-2 antigens and antibodies on the same platform. Unlike traditional rapid tests, the QIAreach platform is able to process around 30 samples an hour and uses a digital detection system that offers customers definitive results in as little as 3 minutes. The QIAreach solution also included the development of Windows software to allow the real-time monitoring of tests and storage of test results.
Project Brief
Recognising that the world was urgently wanting to capture data related to SARS-CoV-2, QIAGEN sought to enhance the QIAreach Software so that test results could be transmitted to Laboratory Information Management and Disease Surveillance Systems. With its focus on quality, the design and development of the solution needed to follow ISO13485, ISO14971 and IEC62304 to maintain quality and manage risk.
Project Activities
With time being of essence and the software release needing to coincide with the product release there was much to do. The Connected Diagnostics team began to quickly embed themselves into QIAGEN processes. Activities included:
• Requirements gathering and market analysis
• Functional design concepts
• Software development plans
• Verification and validation plans
• Risk management
• Development of user manuals
• Training of sales and marketing teams
• Signoff and launch
Project Outcome
The design, development, verification and launch of the QIAreach Software was completed on time and within budget. The functionality of the software was enhanced to not only allow the transmission of test results but also for data suited for epidemiological purposes to be captured. The final product satisfied all processes relating to ISO13485, ISO14971 and IEC62304. The team at Connected Diagnostics continues to work on the enhancement of the QIAreach Software.
Testmonial
“We contacted Chris whilst searching for experts in connectivity related to tuberculosis. His credentials and history spoke for themselves and were exactly what we needed. Chris and the team at Connected Diagnostics have helped to strategically shape and develop software that will be used by QIAGEN customers in the fight against latent tuberculosis and Covid-19. Their input and expertise in this area has been extremely valuable and they have quickly understood our internal quality processes to deliver.” - Dr. Ali Murat Kaya, Senior Digital Innovation Manager at QIAGEN